 Takara Bio is promoting supply of RetroNectin® to clinical development of Engineered T-cell Therapy actively pursued worldwide in recent years, and expects higher sales growth in the future. Under the agreement signed with Takara Bio, Kite is allowed to use RetroNectin® for production of its most advanced product candidate, KTE-C19, a therapy in which a patient’s T cells are genetically engineered to express a CAR designed to target the antigen CD19 for the treatment of B-cell lymphomas and leukemias. By broadening our scientific discovery efforts across new technologies, we are pursuing our goal of developing cell therapies that not only continue to advance safety and efficacy, but are also able to treat a broader range of cancers. And currently, we’re seeking to do even more. Kite is advancing clinical development of several Engineered T-cell Therapy product candidates. We’re proud to be a leader in the treatment of B-cell malignancies. With such advantages, RetroNectin® method is now becoming increasingly standard in protocols utilized for “Engineered T-cell Therapy”, which includes promising TCR and CAR gene therapies recently raising higher expectations. The patented technologies licensed to Kite are based on Takara Bio’s proprietary RetroNectin® method, which includes a technology enabling highly efficient gene transduction to cells by retrovirus vector and expansion of T-cells with high efficiency. Takara Bio now has ten licensees permitted to use RetroNectin® patents commercially. Under this agreement, Takara Bio also provides Kite with reliable supplies of RetroNectin®. 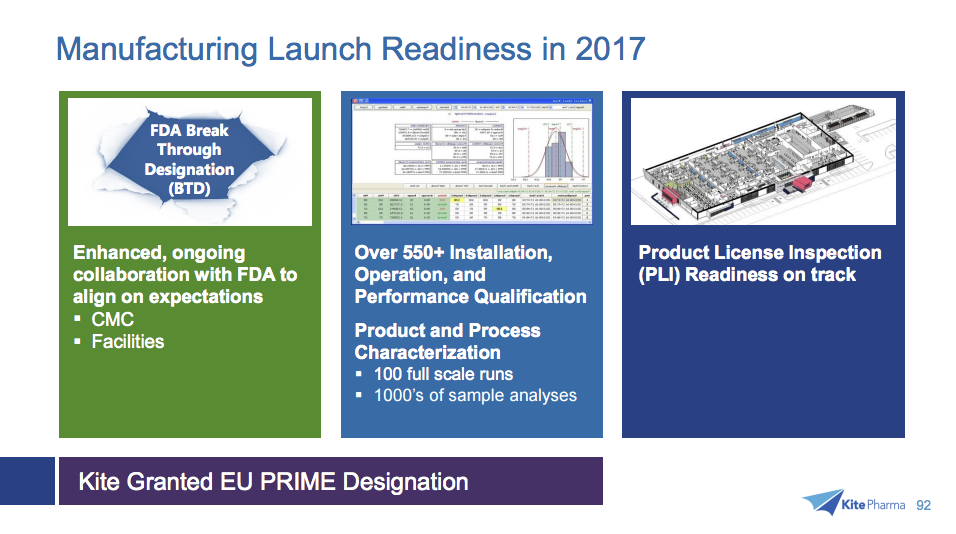
(“Kite”), United States, under which Takara Bio grants Kite a commercial license to use applicable patents relating to RetroNectin®. announces that it has entered into a License and Supply Agreement with Kite Pharma, Inc. Non-GAAP net loss attributable to common stockholders for the third. 
Net loss attributable to common stockholders was 73.9 million, or 1.49 per share, for the third quarter of 2016. 
Kusatsu/Shiga, Japan - J– Takara Bio Inc. General and administrative expenses were 25.0 million for the third quarter of 2016, and includes 10.3 million of non-cash stock-based compensation expense. Factors that could influence actual results include, but are not limited to, economic conditions, especially trends in consumer spending, as well as exchange rate fluctuations, changes in laws and government systems, pressure from competitors’ prices and product strategies, decline in selling power of the Company’s existing and new products, disruptions to production, violations of our intellectual property rights, rapid advances in technology and unfavorable verdicts in major litigation. Actual results may vary significantly from these forecasts due to various factors. While such statements are based on the conclusions of management according to information available at the time of writing, they reflect many assumptions and opinions derived from information that includes major risks and uncertainties. Statements in this news release, other than those based on historical fact, concerning the current plans, prospects, strategies and expectations of the Company and its Group represent forecasts of future results. For more information:PR・IR Department, Takara Bio Inc.Į-mail: article is translated from press release in Japanese for your convenience. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
